Summary
The Netherlands Commission on Genetic Modification (COGEM) is an independent scientific advisory body of the Dutch government with two statutory tasks: 1) to advise the Minister of Infrastructure and Water Management and the ministry’s regulatory bodies and licensing authorities on the possible risks to human health and the environment of work with genetically modified organisms (GMOs), and 2) to inform the government on the ethical and societal aspects of genetic modification.
COGEM’s remit covers all areas of biotechnology, from agriculture to medical applications and from laboratories to the large-scale and commercial introduction of GMOs. COGEM does not give advice on issues relating to food or feed safety or on the possible risks to test subjects and patients in clinical trials or the authorisation of medical products. The Commission consists of no more than 40 members and associated members divided across three subcommittees and is supported by a secretariat of eight people.
Advisory and policy reports in 2025
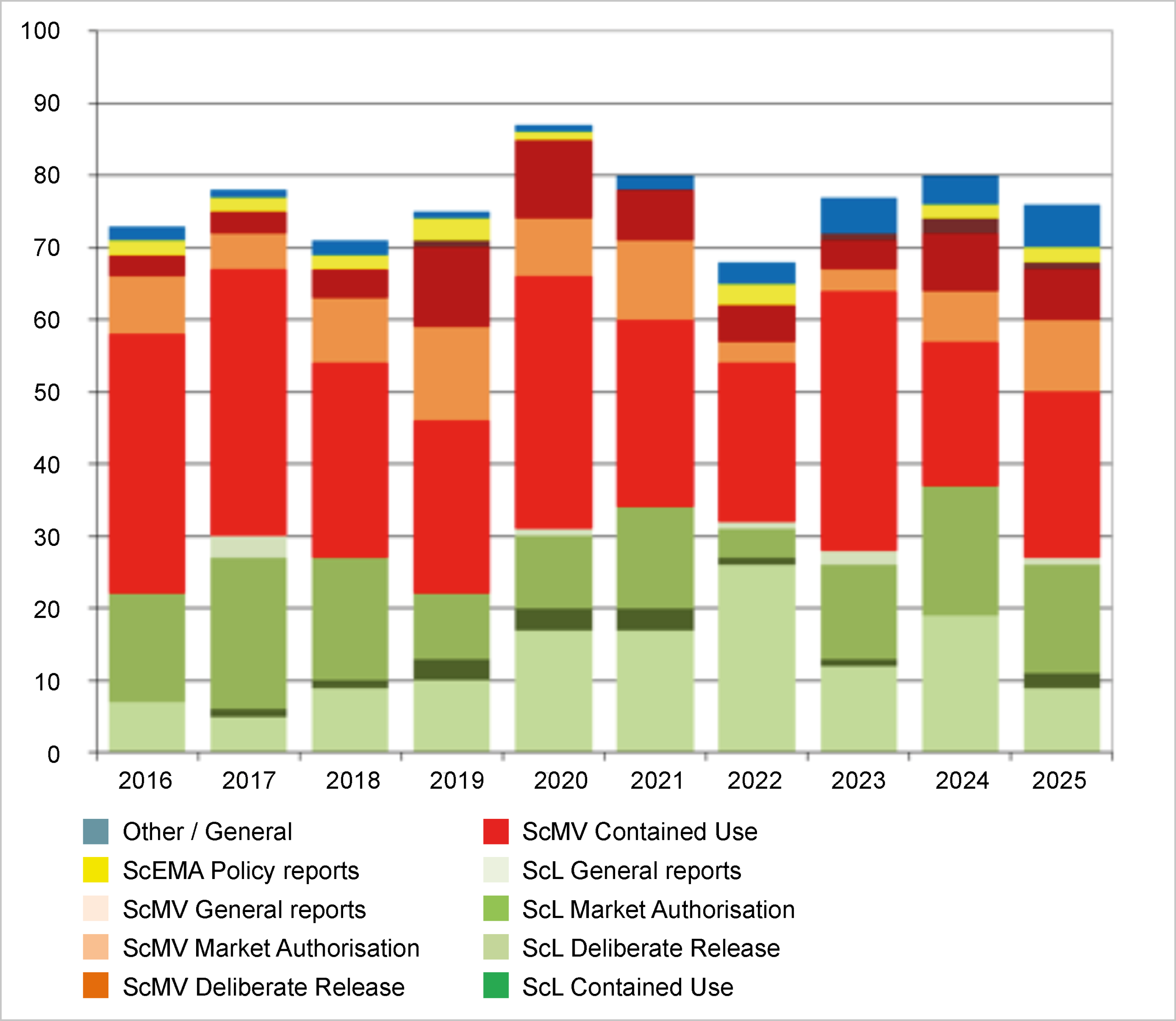
In 2025 COGEM published 76 advisory reports and policy reports. The COGEM subcommittees on Agriculture (ScL) and Medical and Veterinary Aspects (ScMV) prepared 41 and 27 reports, respectively. The subcomitee on Ethics and Societal Aspects (ScEMA) prepared one policy report. Six publications were prepared jointly by two or three subcommittees.
The majority of publications (83%) concerned solicited opinions, most of which were requested by the GMO Bureau on specific permit applications. In addition, the Ministry of Infrastructure and Water Management requested advice on proposals to amend the GMO regulations. The unsolicited advice generally concerned refinements and clarifications of previously issued advice with generic environmental risk assessments for clinical studies and work with viral replicons. Unsolicited advice was also provided on several occasions on amendments to the Dutch GMO Regulation. Furthermore, for the first time in years, a permit was requested for a field trial with a genetically modified crop (GM potatoes) in the Netherlands.
Two policy reports were published in 2025. The first report investigates whether the dynamics of the public debate on genetic modification in the Netherlands have changed in recent years. The main conclusions of this study were that there is no longer a broad public debate. The debate has become institutionalized, with stakeholders as participants, and mainly concerns genetic modification in agriculture. The tone of the debate and the arguments used have hardly changed over time in this institutionalized debate. However, stakeholders who are critical of genetic modification perceive the Dutch government as less neutral or even no longer neutral in the debate because the government agrees with the European Commission's proposal to exempt so-called NGT1 plants from GMO regulations.
The second publication concerned a joint open letter from the German Zentrale Kommission für die Biologische Sicherheit (ZKBS) and COGEM to the European Commission. In this letter, both organizations expressed their concerns about the authorization of medicines containing so-called self-amplifying mRNAs (samRNAs) or viral replicon particles (VRPs). Both commissions believe that an adequate environmental risk assessment of medicines based on viral replicons and samRNAs is crucial, both to assess the risks and to ensure confidence in and acceptance of medicines developed using modern biotechnology.
COGEM Meetings
Over the past year, COGEM organized two meetings: a closed workshop for its European sister organizations and its anniversary symposium aimed at a broad audience of scientists, policy officials, other interested parties, and current and former members and employees of COGEM itself.
The workshop focused on the issues surrounding samRNAs and viral replicons. Together with a number of its European counterparts, COGEM explored the opportunities offered by these technologies, the potential risks and legal implications.
During the anniversary symposium marking COGEM's 50th anniversary, we not only looked back at what genetic modification and biotechnology have brought us, but above all looked ahead to the future possibilities and challenges that biotechnology offers. The promising new developments in biotechnology offer opportunities for a more sustainable world, better healthcare, and higher food production. However, they also lead to new challenges and questions, including those concerning regulation, licensing procedures, and safety. Several speakers pointed out that the innovation ecosystem in the Netherlands and the EU is not in order. An integrated approach by the government to biotechnology is necessary to realize the opportunities for society. With its cabinet vision on biotechnology, the Dutch government has taken a first step to address this, but much depends on further detailing and implementation.
Research reports
COGEM commissions external research to support its advisory and monitoring tasks. In 2025, a number of research projects were started or carried out. Two of the research reports were published last year. Four other research reports were completed in 2025, but have not yet been published. These reports will be published in the course of 2026.
The first published research report ‘Grey Areas in the Regulation of Green and Red Biotechnology. Towards robust legislation for rapid technological change’ provides building blocks for more resilient legislation in the field of GMOs.
The second research report ‘Introgression from cultivated plants into their wild relatives’ is a literature review to gain insight into whether introgression between cultivated plants and wild relatives occurs, how it can be detected, the factors that contribute to it, the scale of its occurrence and its potential impact on wild populations.
Both research reports are available in English and can be downloaded from the COGEM website.
COGEM Members
Three members stood down in 2025: Emeritus prof Johan Memelink (Leiden University), Lonneke Poort LLM PhD (Erasmus School of Law), and Rommie van der Weide PhD (Wageningen-UR).
COGEM was pleased to welcome one new (associated) member in 2025, Jan Wisse MSc (Nederlandse Zuivel Organisatie).
